Welcome to Dongguan Yinso Medical Packaging Co.,Ltd.
Welcome to Dongguan Yinso Medical Packaging Co.,Ltd.

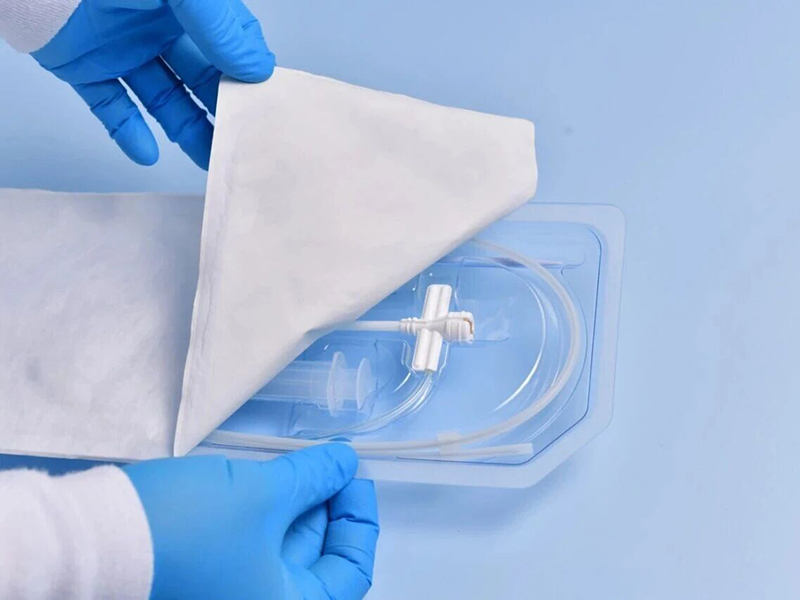
Ethylene Oxide (EO)
Gamma Irradiation
Hydrogen Peroxide Plasma
Steam Sterilization
ISO 11607 series (Umbrella standards)
Part 1: Requirements for materials, sterile barrier systems and packaging systems
Part 2: Validation requirements for forming, sealing and assembly processes
EN 868 series (European standards)
ASTM series (American Society for Testing and Materials) – Examples of common method standards:
ASTM F1980-21: Standard Guide for Accelerated Aging of Sterile Barrier Systems and Medical Devices
ASTM F88/F88M-23: Standard Test Method for Seal Strength of Flexible Barrier Materials
ASTM F1929-23: Standard Test Method for Detection of Leaks in Porous Medical Packaging (Dye Penetration)
GB/T 19633 (Adoption of ISO 11607) — Now updated into two parts:
GB/T 19633.1-2024 (ISO 11607-1)
GB/T 19633.2-2024 (ISO 11607-2)
YY/T 0681 series (Adoption of ASTM test methods)
YY/T 0698 series (Adoption of relevant material standards)
Tyvek®
PE/PET composite films
Thermoformed blister trays
| Test Item | Chinese Standard | International Standard |
|---|---|---|
| Accelerated Aging (Stability Testing) | YY/T 0681.1-2018 | ASTM F1980-21 |
| Transport Simulation | YY/T 0681.15-2019 | ASTM D4169-23e1 |
| Visual Inspection | YY/T 0681.11-2014 | ASTM F1886/F1886M-25 |
| Heat Seal Strength | YY/T 0681.2-2022 | ASTM F1898-15 |
| Seal Integrity (Dye Penetration) | YY/T 0681.3-2010 | ASTM F1140/F1140M-13(2025) |
| Bubble Test for Gross Leaks (Internal Pressurization) | YY/T 0681.4-2021 | ASTM F2096-11 |
| Microbial Barrier Testing (Airborne) | YY/T 0681.10-2011 | ASTM F1608-21 |
| Microbial Barrier Testing (Additional) | YY/T 0681.14-2018 | DIN 58953-6:2025 |
info@yinsopack.com
+86 15014837000(Wechat/WhatsApp/Skype)
NO.59 Meilin Road, Dalingshan Town, Dongguan City,Guangdong Province, China
Get a Free Prototype + Compliance Report Within 5 Days

Yinso Packing has been a leading figure in the medical packaging industry since 2007. We are committed to providing reliable, cost - effective, and innovative rigid thermoformed packaging solutions to top medical companies.
© 2025 Dongguan Yinso Medical Packaging Co.,Ltd All Rights Reserved.
AddressNO.59 Meilin Road, Dalingshan Town, Dongguan City,Guangdong Province, China
Emailinfo@yinsopack.com
Tel(Wechat/WhatsApp/Skype)+86 15014837000
About Us Medical Blister Packs Products Capabilities Blogs Contact Us